
Migraine? CEFALY® can help you!
Try CEFALY® Now-
Drug Free Pain Relief
-
Clinically Proven
-
One Device, Two Settings
-
Treatment & Prevention

Experience a Drug Free Migraine Treatment Today
CEFALY is the #1 FDA cleared drug free migraine treatment that can help improve your quality of life and free you from migraine pain. With no prescription needed, this simple-to-use medical device works to treat acute migraine attacks quickly, prevent future episodes, and may cut your number of migraine days by almost half.
With CEFALY you can live your life without darkened rooms or the forgetfulness, grogginess, and other bothersome side effects that come with some medications. Give yourself the confidence you need to make plans and keep them, without worrying about your migraines.
CEFALY Featured In Australian News
-
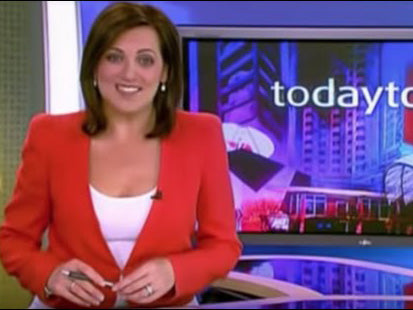
New Hope for Migraine Sufferers on Today Tonight

Cefaly Device on A Current Affair

Cefaly Today Tonight Story

Migraine Device Today Tonight Story

NEW Drug Free Solution for Migraines & Headaches 2017
1 / of 5ReviewsReal People. Real Results
-

I have suffered from migraines all my life, a good friend recommended Cefaly and I am so thankful. My migraines are now less intense and less frequent. Now when one comes along I just use Cefaly and carry on with my day as normal!
Emily
-

Cefaly has saved my life. I was suffering so much with daily, intense migraine attacks. I could not even leave my room for days on end. Ever since my first use of Cefaly, I have not had one attack since. Its been about 6 years now if using Cefaly daily and I am migraine free.
Gypsy
-

Pleasantly surprised at how effective this is. I use the attack mode most days and find it brings down my attack 1-2 on the pain scale. I then use the preventative mode each night before bed and have started to notice a decrease in the number of breakthrough attacks.
Natalie
-
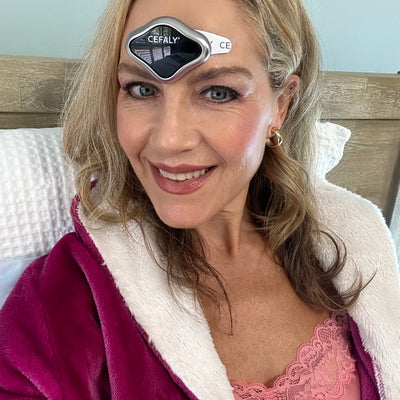
Highly recommend for anyone to try it as part of their toolbox in managing migraine. Cefaly had completely changed my life. I am now able to have control and stick to my commitments and even make plans! The pain reduction is amazing and my breakthrough attacks have reduced by 80%, couldn't be happier!
Sandra
-
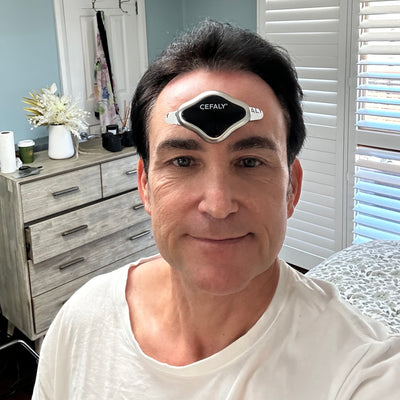
I have always suffered with tension headaches for as long as I can remember. Cefaly has reduced them significantly from even coming on, and when they do just one treatment with Cefaly completely eradicates it. I was concerned about the price at first, but realised I have saved a lot of money after purchasing the Cefaly.
Simon
-

Cefaly has really changed my life. I used to suffer from severe migraines but I’m so pleasantly surprised how much this device has helped me!! I’ve tried many different treatments but this has been the only one that has worked for me. I use it at the first sight of a headache and it works like magic every time. Highly recommend!
Tanisha
-
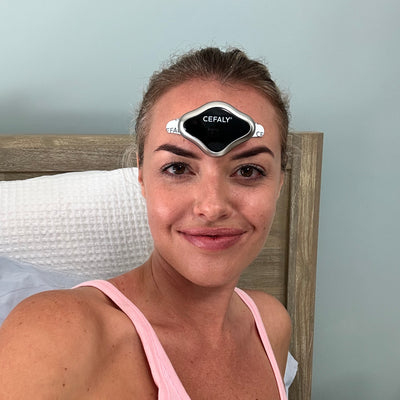
I use my Cefaly every night before going to sleep and it is a significant help in reducing the number of migraines and also helps me get to sleep. It's now part of my daily toolbox.
Natalie
Trusted to relieve migraine pain since 2008
CEFALY has treated more than 2 billion migraine attacks to date. Multiple clinical trials have shown that CEFALY is an effective, safe and drug-free migraine treatment with minimal side effects.
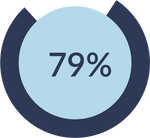
of CEFALY users saw migraine pain relief following one hour of ACUTE treatment. 32% of CEFALY users saw pain freedom.
Read Migraine Treatment Study


of CEFALY users saw at least a 50% reduction in migraine days following daily use of PREVENT treatment.
Read Migraine Treatment Study
 4.7Rated 4.7 out of 5 starsBased on 163 reviews5Rated out of 5 stars124Total 5 star reviews: 1244Rated out of 5 stars36Total 4 star reviews: 363Rated out of 5 stars3Total 3 star reviews: 32Rated out of 5 stars0Total 2 star reviews: 01Rated out of 5 stars0Total 1 star reviews: 098%would recommend this productSlide 1 selected163 reviews
4.7Rated 4.7 out of 5 starsBased on 163 reviews5Rated out of 5 stars124Total 5 star reviews: 1244Rated out of 5 stars36Total 4 star reviews: 363Rated out of 5 stars3Total 3 star reviews: 32Rated out of 5 stars0Total 2 star reviews: 01Rated out of 5 stars0Total 1 star reviews: 098%would recommend this productSlide 1 selected163 reviews- SJSharon J.Verified BuyerI recommend this productRated 5 out of 5 stars26 minutes agoFinally, some relief!
I had continuous migraines for months. I tried the Cefaly and within a couple of weeks, things had improved significantly. I would highly recommend this for anyone in the same boat
Was this helpful?CACefaly Australia15 seconds agoHi Sharon,
Thank you for your wonderful feedback! We’re so glad CEFALY Enhanced has helped bring you relief. Your recommendation means a lot to us.
Kind regards,
OzHealth Pharma Team - AFAnna F.Verified BuyerI recommend this productRated 5 out of 5 stars3 months agoFantastic!
I didn't expect results so quickly!
I have only been using Cafaly for 2 weeks and already my migraines have reduced. The previous 2 weeks I experienced migraine on 10 of 14 days. The fortnight using Cefaly I have had 5 days with migraine.
Was this helpful?CACefaly Australia3 months agoHi Anna,
Thanks for sharing your fantastic results. We're thrilled to hear CEFALY Enhanced is making such a significant difference so quickly.
Kind regards,
OzHealth Pharma Team - CSCassandra S.Verified BuyerI recommend this productRated 5 out of 5 stars3 months agoIt helps
It’s only been 3 weeks.
It definitely helps reduce frequency and duration.
Perhaps more simple details in how to change the intensity would be helpful
Was this helpful?CACefaly Australia3 months agoHi Cassandra,
Thanks for sharing your experience! We're thrilled CEFALY Enhanced is helping reduce the frequency and duration of your episodes.
Kind regards,
OzHealth Pharma Team - RRobynI recommend this productRated 5 out of 5 stars3 months ago5 Stars
Very helpful for pain relief
Was this helpful? - SZsamantha Z.Verified BuyerI recommend this productRated 5 out of 5 stars4 months agoFantastic for managing symptoms
In my experience with the Cefaly, I found it reduced pain, dizzyness and nausea or ended a migraine early. Cannot recommend it enough to any chronic sufferers.
Was this helpful?CACefaly Australia3 months agoHi Samantha,
Thank you! We’re thrilled CEFALY Enhanced is helping you and appreciate you recommending it to others!
Kind regards,
OzHealth Pharma Team
Loading...Loading...- Choosing a selection results in a full page refresh.
- Opens in a new window.





















